|
3/18/2024 0 Comments Bohr model of atoms Electrons can jump from one orbit to another by emitting or absorbing energy. The orbits are labeled by an integer, the quantum number n. Rutherford has given a theory of the structure of atoms. 1-25 1 In order to explain the results of experiments on scattering of rays by matter Prof. Instead of allowing for continuous values for the angular momentum, energy, and orbit radius, Bohr assumed that only discrete values for these could occur (actually, quantizing any one of these would imply that the other two are also quantized). Bohr model of the atom In the Bohr model of the atom, electrons travel in defined circular orbits around the nucleus. Bohr Model of the Atom On the Constitution of Atoms and Molecules Niels Bohr Philosophical Magazine Series 6, Volume 26 July 1913, p. Around 400 BC, a Greek philosopher named Democri. The absolute value of the energy difference is used, since frequencies and wavelengths are always positive. Atomic Models: Centuries ago, people didn’t know exactly what was inside an atom, but they had some ideas. In this equation, h is Planck’s constant and E i and E f are the initial and final orbital energies, respectively. In Bohr’s model of the hydrogen atom, one electron, carrying a negative electrical charge, circles a nucleus consisting of a single proton, which has a positive. This observation is connected to the discrete nature of the allowed energies of a quantum mechanical system.\] These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells. It is observed that line spectra discussed in the previous sections show that hydrogen atoms absorb and emit light at only discrete wavelengths. The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus (Figure 1). It does introduce several important features of all models used to describe the distribution of electrons in an atom. Bohr built upon Rutherfords model of the atom. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it does not account for electronelectron interactions in atoms with more than one electron. 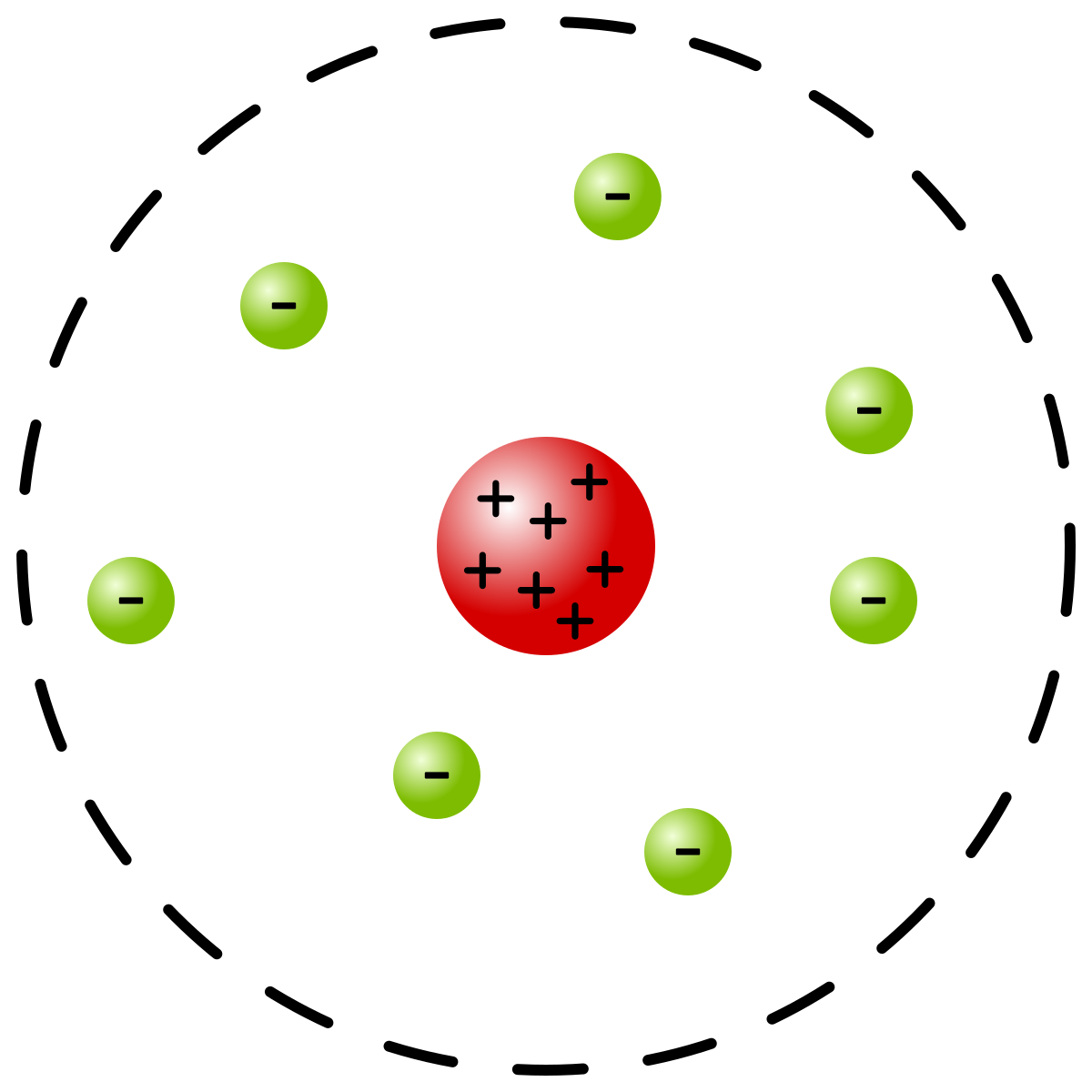  
Atoms are the basic units of chemical elements and were once believed to be the smallest indivisible structures of matter. Bohr thought that electrons orbited the nucleus in circular paths whereas in the modern view atomic electron structure is more like 3D standing waves. How did scientists figure out the structure of atoms without looking at them Try out different models by shooting light at the atom. From this, it is easy to see that like charges (charges of the same sign) repel each other. In 1913, the physicist Niels Bohr introduced a model of the atom that contributed a greater understanding to its structure and quantum mechanics. Bohr model of the hydrogen atom was the first atomic model to successfully explain the radiation spectra of atomic hydrogen. Khan Academy is a nonprofit with the mission of providing a free, world-class education for anyone, anywhere. If \(Q_1\) and \(Q_2\) are the same sign, then the curve which is a purely repulsive potential, i.e., the energy increases monotonically as the charges are brought together and decreases monotonically as they are separated. Learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
